EARLY DRUG DEVELOPMENT & NONCLINICAL SAFETY
Medical Toxicology Integrated with Pathology Insight
ANATOMO-PATHOLOGY & MEDICAL TOXICOLOGY
Pathology and medical toxicology are two sides of the same coin in preclinical and early clinical drug development. When they work together, your program gains clarity, speed and regulatory confidence. Toxicologic Pathology offers that integrated expertise—DABT‑certified toxicology paired with translational pathology—so you can move safer, faster and smarter from discovery to first‑in‑human studies.Why integration mattersEarly identification of target‑organ effects: Pathology reveals tissue and cellular changes; toxicology interprets those changes in the context of dose, kinetics and mechanism. Combined, they distinguish incidental findings from true safety signals.Mechanism‑driven risk assessment: Pathologists characterize lesion morphology and progression; toxicologists map those lesions to exposure, ADME and pharmacology to define mechanism and human relevance.Better translational predictivity: Integrated review reduces surprises when moving to clinical phases by aligning animal pathology with clinical biomarkers and potential adverse outcomes in humans.Faster regulatory acceptance: Regulatory reviewers expect coherent narratives linking histopathology, toxicology data and safety margins. A single, integrated team produces consistent, defendable dossiers and study reports.Expert integrated reports: Concise, scientifically rigorous study narratives that link histopathological findings to toxicology data, pharmacokinetics and mechanism-of-action.Mechanistic investigations: Focused follow‑up studies and targeted assays (IHC, digital pathology quantitation)
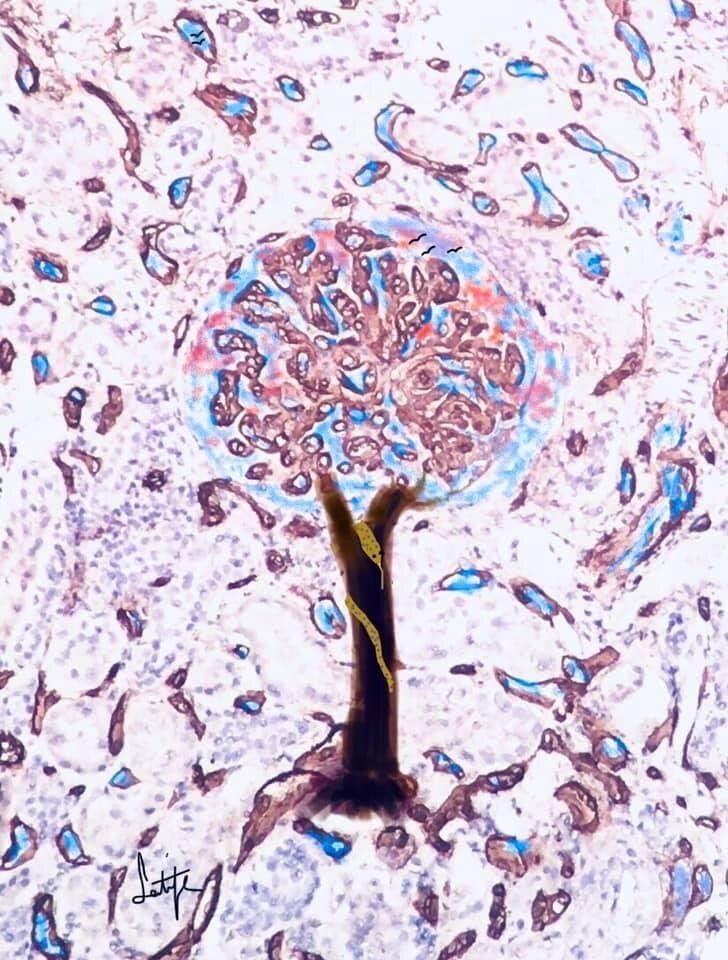
Morphology as a Foundation for Interpretation
What we see is only the beginning.
Pathology reveals patterns—but meaning emerges through interpretation.
Across tissues, species, and systems,
the question is not only what is present,
but what it means for human risk.it stand out
Whatever it is, the way you tell your story online can make all the difference.
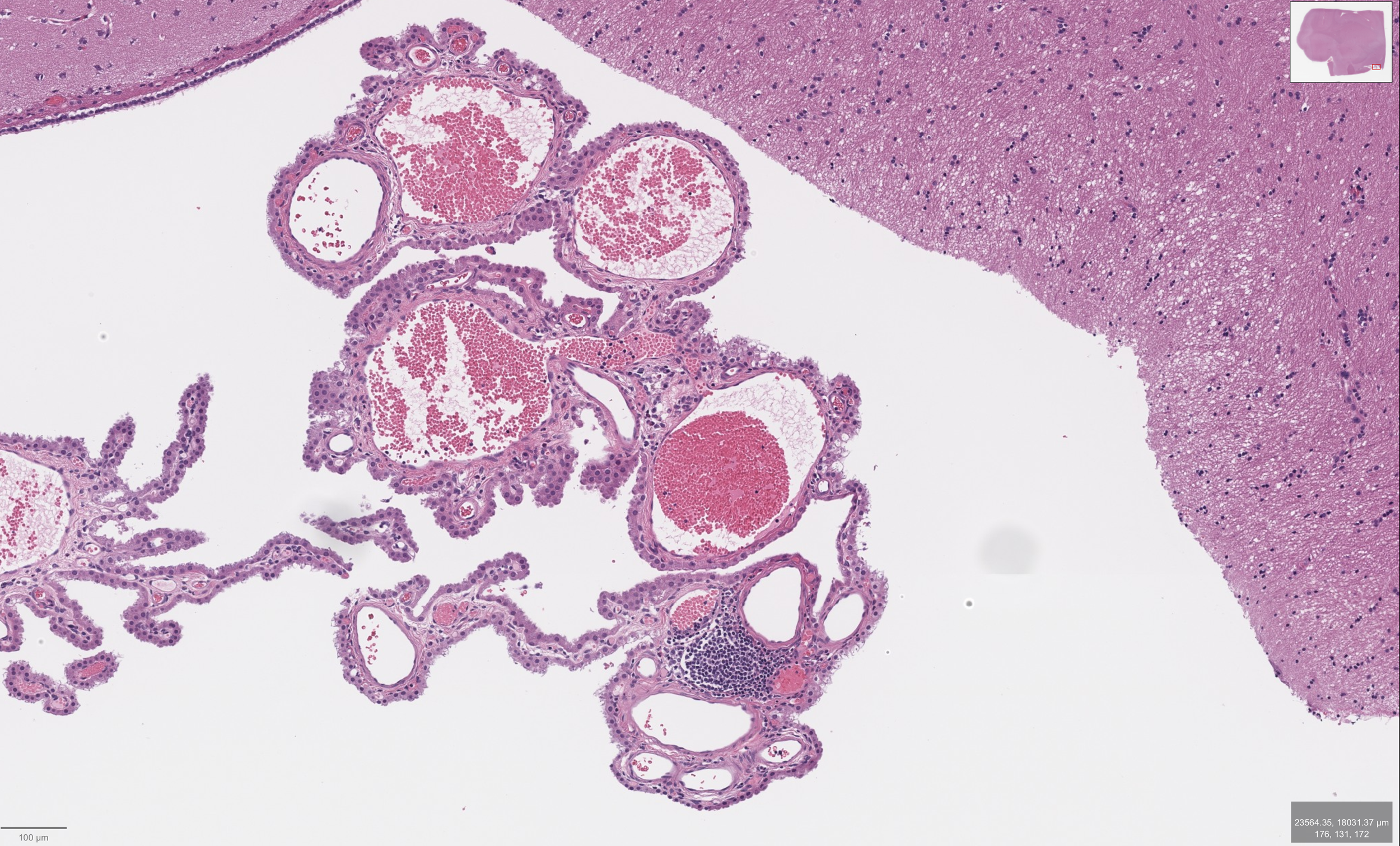
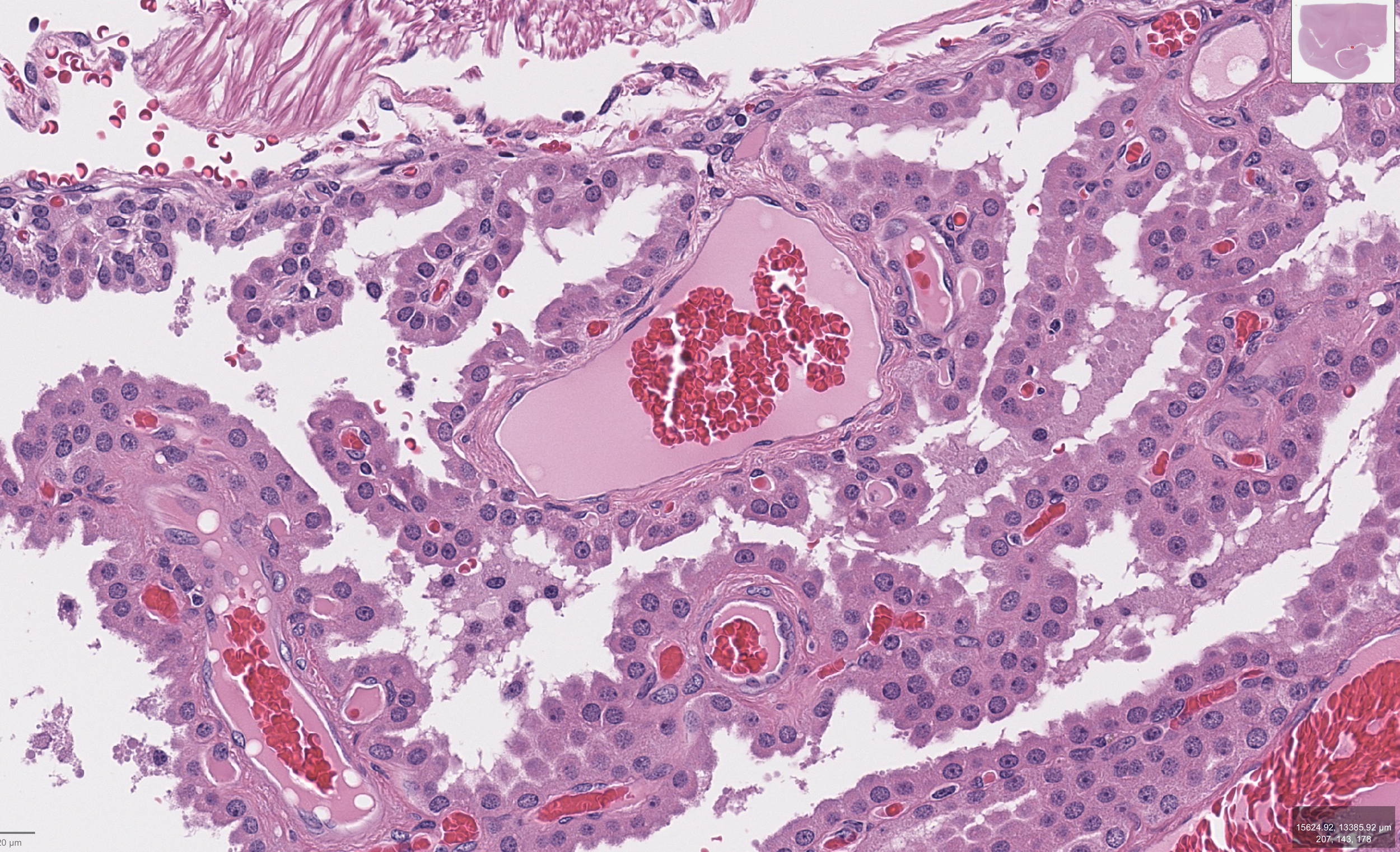
The Organ Behind the Interpretation
The examined tissue shows choroid plexus with preserved, well-organized papillary architecture, characterized by delicate branching fronds containing fibrovascular cores and lined by a single layer of uniform cuboidal to low columnar epithelial cells. The epithelial cells display bland, round nuclei without atypia or mitotic activity, and the overall architecture remains orderly without stratification or complexity. These features are consistent with normal anatomical choroid plexus histology, reflecting intact structural organization and differentiation.
gross pathology & histology
Necropsy Specimens will only achieve their objectives if biopsy/full sampling, collection, fixation & transportation are conducted according to Specific guidelines and/or standard operating procedures.
Understand gross technics in necropsy rooms
manage & control your handling & fixation technics
trimming Guidelines for safety studies
State-of-art Histology procedures
trimming, Embedding & Cutting procedures
Revision & update Histology protocols
Expertise
Oral, Dermal, infusion, inhalation, carcinogenicity
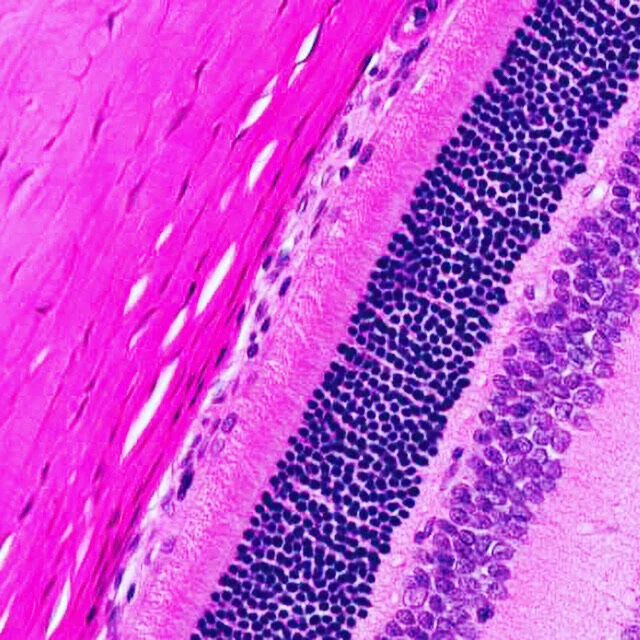
Ocular Pathology
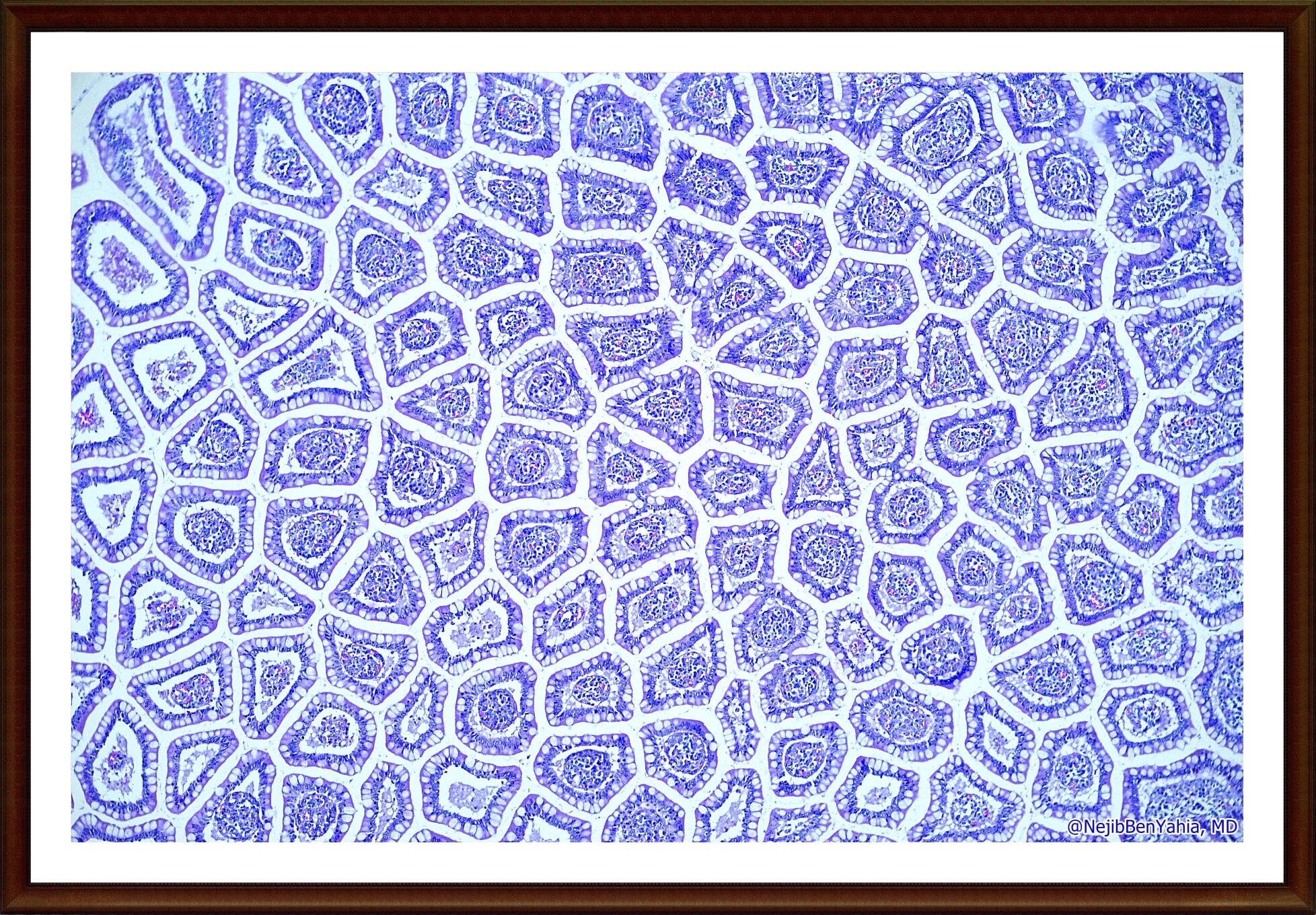
histopathology
Preclinical R&D programs including adequate IND submissions represent a regulatory-driven prerequisite before Authorising the administration of a new drug or implantation of a new medical device. The role of the Toxicological Pathologist is to act as a preclinical expert: He/She diagnoses, interprets and deliver written risk assessments on any organ/tissue change related directly or indirectly to the administration of the new drug. the final pathology report represents the end-result of the in vivo experiments and will be part of the IND submission.
DIGITAL PATHOLOGY
Digital pathology has been widely discussed since several years in Societies of Surgical Pathologists. Recently, it's use has been widely increased in additional fields such as in Academia for educational purposes, but also in Non-Clinical drug development research.
In 2017, the FDA approved for the first time the Whole Slide Imaging Systems for digital pathology that allows the interpretation of digital surgical pathology slides prepared from biopsied tissues. In this non-binding guidance FA describes the details about the technical performance of WSI. Additional Peer-Reviewed publications are coming soon and will be announced to all followers of digital pathology.
Since, 2009 I have been trained within a digital pathology Environment and in 2019, I obtained nSH/DPA certification in digital pathology. Special blog posts are published on a regular base including Podcasts giving you the latest updates about digital pathology within toxicological pathology.
Feel free to send me your questions about digital pathology
Double-board certified toxicologist & pathologist
20-years expertise in early drug development 
regulatory roles & expertise
Designs & Interpret nonclinical safety & efficacy studies
solicits bids to evaluate and select the appropriate contract research Organisations
manages & monitors preclinical and nonclinical studies
serves on project teams, expert panels, & scientific advisory boards
assists in new project development an existing product Defence
reviews & writes expert reports, white papers, study reports, product Labelling, safety data sheets; investigator brochures
acts as a liaison between sponsors and regulatory authorities
provides gap analysis & addresses needs & assessments for existing programs
renders subject matter expertise during due diligence activities
Assists financial forms in evaluation of specific data for small & large research institutes and pharmaceuticals
types of toxicologic programs & IND submissions
core toxicology pathology Programs
TRANSLATIONAL laboratory animal WELFARE
Replace, Reduce, Refine, Respect (4Rs) is an integrated framework that improves animal welfare and scientific reproducibility. Replacing animals with valid alternatives lowers species-related variability. Reducing numbers through robust design and power calculations limits confounders while retaining statistical validity. Refining procedures and husbandry minimizes stress, pain, and handling effects that distort endpoints. Respecting animals via continuous welfare monitoring and transparent reporting enhances data interpretability and study replicability. Centering welfare yields steadier baseline physiology,
reduced inter-animal variability, and improved signal-to-noise in preclinical studies, producing more ethical and reproducible
compliance 21 cfr part 11
Title 21 CFR Part 11 is the FDA regulation that governs electronic records and electronic signatures. It sets the requirements under which electronic records and signatures are considered trustworthy, reliable and equivalent to paper records. In practice, Part 11 applies to pharmaceutical firms, medical device manufacturers, biotechnology companies and, in some cases, GLP units within academic settings.Good Laboratory Practices
in the experimental or translational research area, GLP is a quality system of management controls for laboratories and Organisations who need to ensure the uniformity, consistency, reliability, reproducibility, quality and integrity of products in development for human and animal health through non-clinical safety tests; varying from physiochemical Properties through acute to chronic toxicity tests.pathologist & Good laboratory practices
Understanding the GLP-workflows from accredited Good Laboratory Practice laboratoriesUnderstanding, reviewing & Implementing standard operating procedures (SOP’s)Auditing nonclinical safety studies, histology & pathology proceduresProviding mock inspections in order to prepare for official regulatory authoritiesRevisions of raw data, incl. necropsy proceduresCorrective actions, Root cause analysisNeed some examples: check your QA blog spot !
questions ?

Copyright & Intellectual Property© 2026 DR. Neyens Elizabeth. All rights reserved.All content made available on this website, including but not limited to text, scientific and technical materials, reports, methodologies, graphics, logos, and the overall design, is the property of Dr. Elizabeth Neyens, unless otherwise expressly stated. This content is protected under applicable intellectual property laws, including the Copyright Act and relevant international conventions. The content of this website is provided for informational and non-commercial use only. You may access and view the material for personal or professional reference; however, no part of this website may be reproduced, distributed, modified, transmitted, or used for commercial or competitive purposes without the prior written consent. The information presented on this website reflects professional expertise in pathology and toxicology and is intended solely for general informational purposes. It does not constitute medical advice, diagnostic interpretation, or regulatory or legal guidance. Access to this website does not grant any license or right to use any intellectual property belonging to DR. Elizabeth Neyens or to any third party. Unauthorized use of the website or its content may result in legal action.